Sample collection kits
Are you looking for simple systems to be used by sample collectors, while guaranteeing that the collected samples will be traceable, protected, and intact? Are you looking for a high-quality and responsive kit sourcing service, combined with rigorous traceability?
In collaboration with investigator sites, Cell&Co designs your sample collection kits and related documentation to meet your specific needs. They offer maximum ease of use and help maximize the quality of your biological samples. The kits may be dedicated to one visit, one patient, one dose, or one cycle, and can be adapted to all types of sample.
In addition to design, we also produce the kits in-house and manage their national and international distribution. Investigator sites are automatically notified when kits are reaching the end of their shelf-life, and Cell&Co manages any necessary resupply. Depending on the project’s constraints, each kit can be provided with a unique identification number for full traceability.
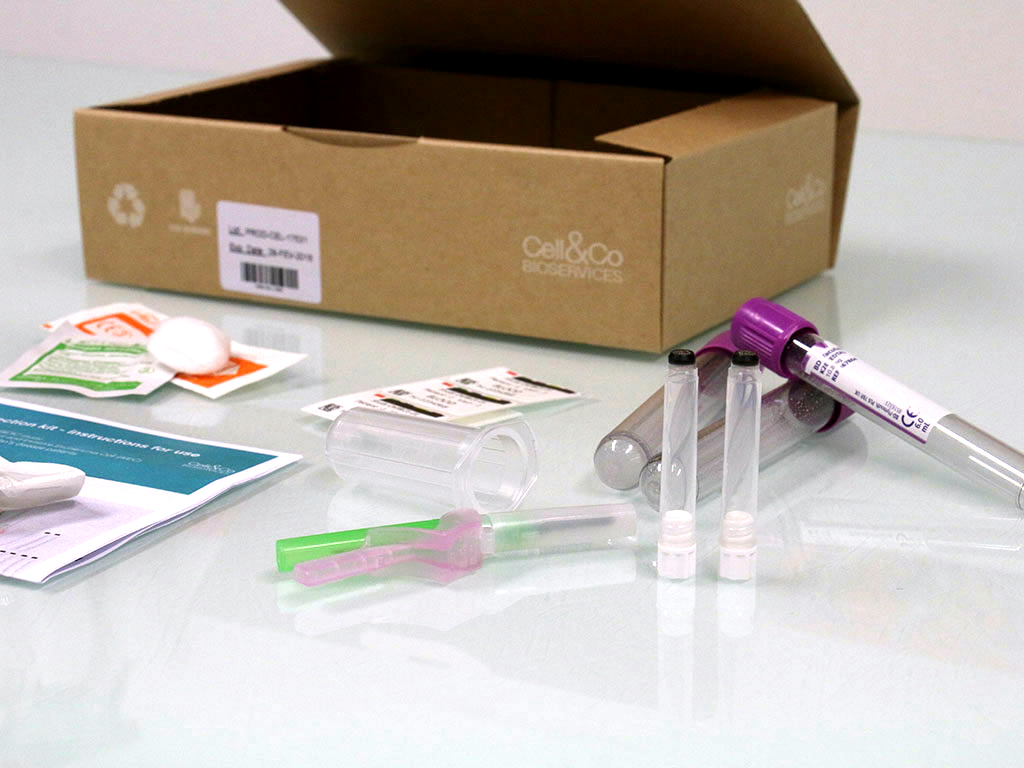
Kits design and production
Step 1
- Need analysis
- Drafting of specifications
Step 2
- Kit design
- Selection and validation of components
Step 3
- Writting of related documents
Step 4
- Provision of demonstration kits
Step 5
- Production, inspection and storage
- Distribution to investigator sites
Step 6
- Disposal & Disassembly